
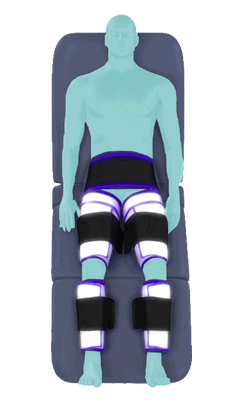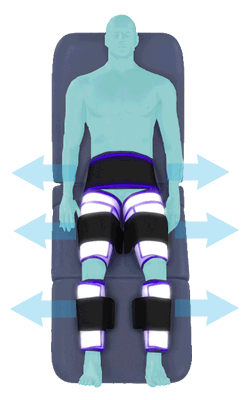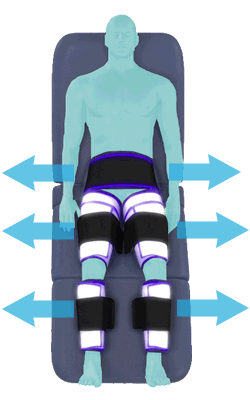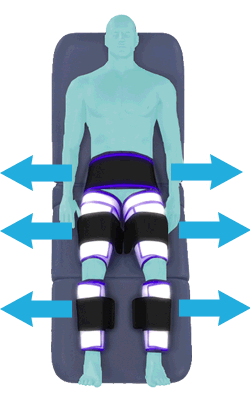
The Renew Cerezen™ system administers counterpulsation therapy. It does this by rapidly inflating/deflating air bladders contained in cloth cuffs. The cuffs are velcroed around each calf, each thigh and the buttocks/waist (a total of 5 bladders).
When inflated, the air bladders squeeze that part of the body, propelling blood from the arteries and veins in that region towards the central body / torso.
The bladders are inflated in a rapid sequence – calf to thigh to buttocks – moving blood distally to proximally.
Bladder inflation and deflation is synchronized with the patient’s ECG signal so that inflation occurs only during the diastolic portion of the heart cycle, i.e. while the heart is resting. The bladders are deflated before each heart contraction, allowing the heart to expel blood into a relatively emptied lower extremity vascular bed, reducing cardiac afterload.
A typical treatment regime consists of 35 one-hour treatments over the course of 7-12 weeks, i.e. 3-5x per week. This is typically followed by ongoing maintenance treatments twice weekly.

Inflatable air cuffs are wrapped around calves, thighs and hips.
Between heartbeats, the cuffs inflate sequentially from the calves up to the hips.
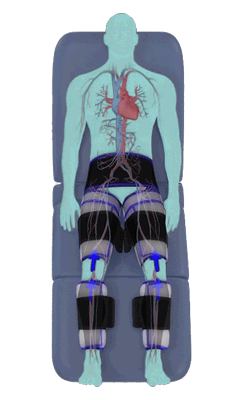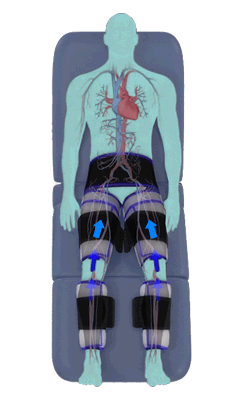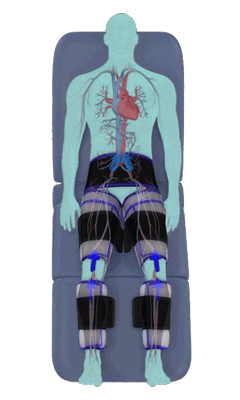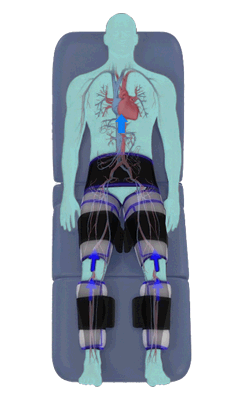
Cuff inflation compresses the blood vessels, forcing blood back to the core body. The rapid to-and-fro motion of blood is interpreted by the body as similar to exercise. This results in release of a number of chemicals such as nitric oxide which increase blood vessel diameter and reduce vascular inflammation.
The cuffs deflate simultaneously prior to the next heartbeat.
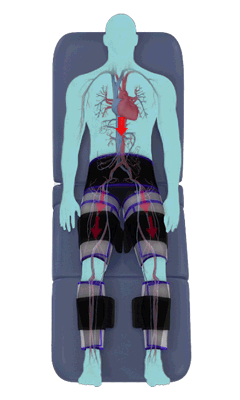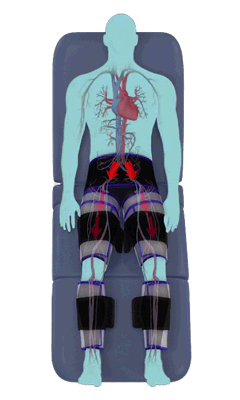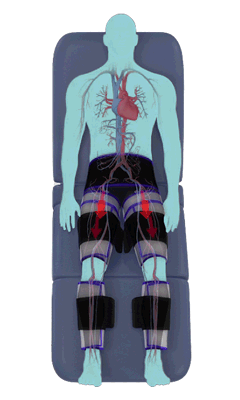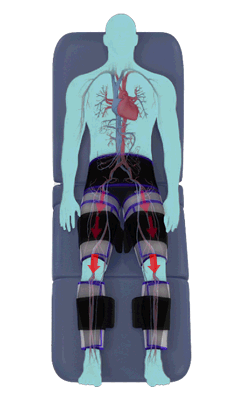
The rapid deflation of the cuffs significantly reduces the amount of work required of the heart to pump oxygenated blood to the rest of the body.
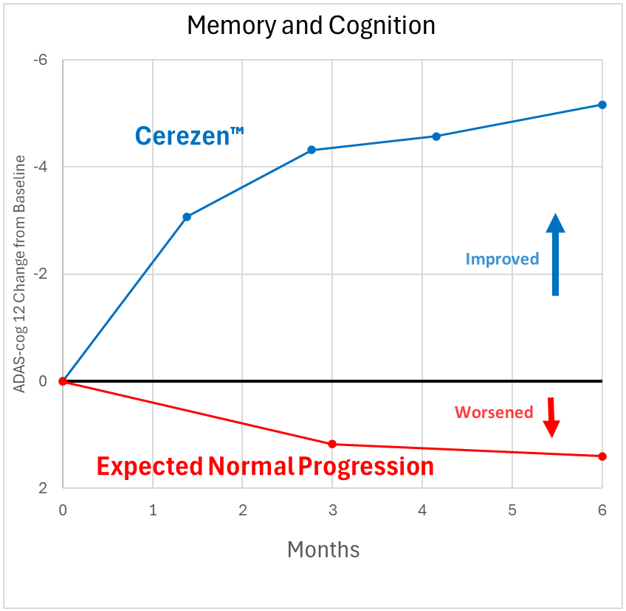
Results of pivotal trial of Cerezen™ in 190 subjects with mild cognitive impairment (MCI) or Alzheimer’s disease.

Cerezen™ is a CE-marked Class IIa medical device certified under the EU Medical Device Regulation (MDR 2017/745).
Certification issued by BSI (2797), Certificate No. MDR 818385
Indication: Cerezen™ is indicated for the treatment of mild cognitive impairment due to Alzheimer’s disease or mild Alzheimer’s disease.
Intended Use: Cerezen™ is intended for use as a component in the overall management of symptoms of cognitive and/or functional impairment experienced by adults with mild cognitive impairment due to Alzheimer’s disease or mild Alzheimer’s disease. It is intended for use under the oversight of a healthcare professional.
Intended Patient Population: Cerezen™ is intended for use in adults suffering from mild cognitive impairment due to Alzheimer’s disease or mild Alzheimer’s disease.
Cerezen™ is CE-marked in the European Union. It is not cleared by the U.S. Food and Drug Administration and is not available for sale in the United States.
The following documents are for trained medical professionals only: